Cell biology experiments are traditionally performed in bulk cell suspensions where cell secretions become diluted and mixed in a contiguous sample. Novel microfluidic methods offer strategies for the encapsulation of cells in aqueous droplets and hydrogels that can be produced at kilohertz frequencies, therefore creating the potential for millions of single cell experiments run in parallel. In fact, the encapsulation of single cell in highly monodisperse small picoliter-volumes spaced by a continuous oil phase, provides isolation of each cell, thus creating the condition where cell qualities are highlighted within the space of individual microreactors and signals can be concentrated to measurable levels otherwise undetectable. Microfluidics methods of cell confinement can be also coupled with fluorescence-activated cell sorting (FACS) for high-throughput microdroplet screening and downstream sensing.
In a single word, yes! Emulsion droplet size can impact how flavors and aromas are released from emulsions such as hot chocolate and mayonnaise. Size also impacts the speed at which drugs or agrochemicals are delivered and released, and the length of time needed between treatments.
Microfluidics provides a tool to manipulate liquids, gases, droplets, cells and particles within micro-channel geometries. The generation of droplets involves controlling the jetting to dripping transition when liquid droplets are pushed into a carrier fluid via a specific chip geometry. The droplets are stabilized using surfactants to avoid coagulation and separation.
Among its various advantages, microfluidic technology has the ability to create three-dimensional flow patterns that achieve precise control over immiscible and miscible fluid mixing.
Droplet-based microfluidic systems have shown unparalleled advantages for the encapsulation of proteins, enzymes, bacteria or viruses. By precisely controlling the formation of the droplets, we can produce hydrogel beads or double emulsion droplets with well-defined sizes, shapes and morphologies, which can then be sorted on conventional Fluorescence Activated Cell Sorting (FACS) machines.

Double emulsion (DE) droplet technology enables useful applications: in vitro evolution (e.g. of viruses); directed evolution of proteins from expression libraries, such as therapeutic proteins, DNA/RNA polymerases with novel or modified activities, or industrial enzymes.
Microfluidic droplets can be sorted on conventional Fluorescence Activated Cell Sorting (FACS) machines, by turning them into double emulsion droplets (i.e. water droplets in an oil ‘shell’, in an aqueous continuous phase).
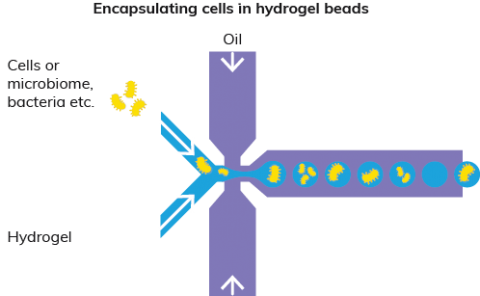
Hydrogels have the unique capability to retain particles or cells of a certain size as well as being porous at the same time, allowing smaller molecules or buffers to diffuse in and out of the hydrogel. Most of them can be kept liquid or harden at temperatures that are compatible when working with cells or bacteria, requiring temperatures most commonly between 4 and 37˚C. The microfluidic method makes it possible to generate hydrogel droplets from a liquid stream of hydrogel that can be hardened into solid hydrogel beads. The use of hydrogel beads offers promising delivery systems for encapsulation and release of proteins, enzymes, bacteria and viruses. Dolomite microfluidic technologies can be used for bead formation with hydrogels such as agarose, alginate, collagen (e.g. Matrigel) and gelatine. Gel beads containing cells can be FACS sorted.

Dolomite’s µEncapsulator Systems are the perfect solution for these applications as they utilize microfluidic methods to directly generate monodisperse droplets, ready for further processing. They can be used to generate droplets ranging in size from 12 to 65 µm, resulting in formulations such as water in oil in water (w/o/w) or hydrogel droplets.
The microfluidic production of a double emulsion on the µEncapsulator System using a 30µm µEncapsulator 1- 2 Reagent Droplet Chip.
Encapsulation of Agarose and Matrigel on the µEncapsulator System using a 50 µm µEncapsulator 1- 2 Reagent Droplet Chip.

Read about how scientists approached the functional cell-based screening by co-encapsulation of secretor yeast cells and reporter mammalian cells in agarose-containing microdroplets.

Microfluidic technology offers a platform for enhancing biological research, allowing the encapsulation of biological cells and molecules in nano or pico-litre volumes. Read our Expert's review of the state of the art of microfluidics-based FACS applications.

Read about how scientists employed droplet-based optical polymerase sorting (DrOPS) to expand polymerase function which uses an optical sensor to monitor polymerase activity inside the microfluidic droplet.

Read about how scientists came up with novel approach for 3D cell culture based on a flow-focusing microfluidic system that encapsulates epithelial cells in Matrigel beads.

Read how scientists use the double emulsion method to create an emulsion suitable for quantitative analysis and sorting in a commercial flow cytometer.

The user-friendly, high-throughput systems offer simple, straightforward encapsulation of cells, bacteria, microbiome, and/or functionalised beads in high precision, identically-sized picolitre droplets.