Alginate is the most abundant marine biopolymer, typically is extracted from seaweed which makes it a naturally occurring anionic polysaccharide. It possesses benefits of low toxicity and is produced at a relatively low cost, making it a popular material for pharmaceutical and biomedical applications. Alginate has been widely used in several applications for example: delivery of small chemical drugs, protein delivery since it minimises denaturation, wound dressings and cell culture.
Microfluidic technology adaptation for alginate synthesis has seen a significant growth in varied application fields. Benefits of reproducibility, real-time control and reduction of waste are few of many factors users are choosing to switch from conventional batch methods to microfluidics.
Drug delivery, wound healing, cell and tissue culture all require inert biomaterials capable of catering to their different requirements. Alginate has inherent structural similarities to extracellular matrices of living tissues making it a model system for mammalian cell culture and tissue engineering, i.e. organ and tissue replacement during organ or tissue failure. Another advantage of using alginate is its mild gelation requirements which can be performed using various methods.
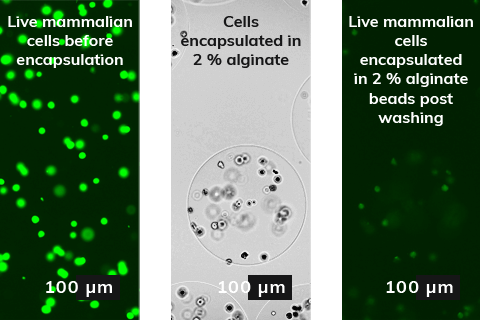
Our application note showcases the benefits of microfluidic technology for mammalian cells encapsulation in alginate beads. The picture on the left summarises the experiment results, the alginate encapsulation and the downstream washing and crosslinking did not harm the mammalian cells. Live cells were visible in the alginate beads. Therefore, microfluidic methods can easily be adapted for creating microreactors and or other sensitive active agents requiring mild gelation and encapsulation.
In a single word, yes! The porous alginate structure allows growth materials transferred and exchanged via the nanopores in the bead structure. Bead size can impact the speed of cell growth and the degradation speed at which drugs or agrochemicals are delivered and released, impacting the length of time needed between treatments.
Microfluidics provides a tool to manipulate liquids, gases, droplets, cell and particles within micro-channel geometries. The generation of droplets involves controlling the jetting to dripping transition when liquid droplets are pushed into a carrier fluid via a specific chip geometry. The droplets are stabilized using surfactants to avoid coagulation and separation.
Among its various advantages, microfluidic technology has the ability to create three-dimensional flow patterns that achieve precise control over immiscible and miscible fluid mixing.
Droplet-based microfluidic systems have shown exceptional advantages for the synthesis of monomer- or polymer-containing droplets inside which the hydrogel structures can be formed using various chemical methods. By precisely controlling the formation of the bead, we can produce alginate particles with well-defined sizes, shapes and morphologies.
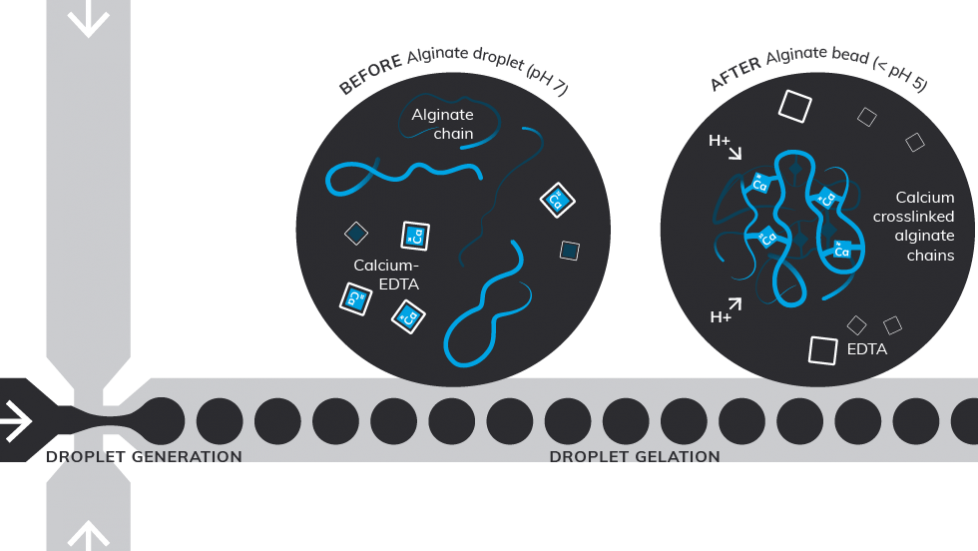
The schematics below showcases the flow focusing microfluidic method example from our application note. The before and after processes of alginate crosslinking using acetic acid in collection phase and how during the droplet gelation the Calcium-EDTA complex dissociates and releases Ca2+ ions to crosslink alginate chains.

Dolomite’s Single Emulsion Systems are the perfect solution for this application as they utilize microfluidic methods to directly generate monodisperse droplets or emulsions, eliminating the need for further processing. They can be used to generate droplets ranging in size from 2 to 200 µm, resulting in formulations such as water in oil (w/o) or oil in water (o/w) emulsions.
Methodology for generation of monodisperse alginate beads in sizes ranging from 80 to 120 µm using microfluidic flow focusing method.

Read the latest featured story about how our experts are using microfluidics to produce small (80-120 µm) alginate beads suitable for cell encapsulation.
Read more about the emulsion stabilizer used in our experiments with alginate. FluoSurf is an emulsion stabiliser for aqueous droplets in fluorinated oil. It is a block copolymer that stabilises emulsions due to its ambiphilic polymer blocks, that allow it to stabilise the interface between aqueous and fluorinated solutions.

Methodology for monodisperse agarose particle production using microfluidic droplet methods, with particle sizes varied between 20 µm to 130 µm.